 |
| Click to Enlarge |
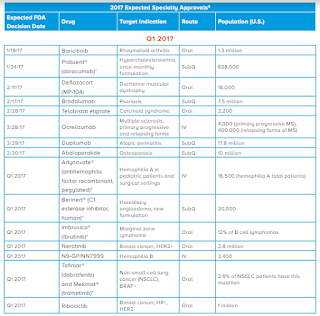
The report offers insight on developments in emerging specialty therapies. Last year, the FDA Center for Drug Evaluation and Research (CDER) approved 22 novel new drugs. While there was a reduction in novel new drugs compared to previous years, there were several expanded indications for previously approved therapies that allowed these drugs to reach additional patient populations. New approvals included a mix of oncology, immunology, rare diseases, and other disease states.
Although the quantity of novel new drugs was lower in 2016, important new drugs became available in each of the broad disease states covered by specialty pharmacy. Specialty drug products accounted for approximately half of new drugs and biologics approved in 2016, similarly to in 2015. Nine specialty agents were approved for rare diseases.
“The specialty pharmacy industry continues to show exceptional growth,” said Paul Urick, Diplomat’s president. “The development of new drugs, as well as expanded indications for previously approved treatments continues to make the robust specialty drug pipeline one of the major drivers of growth.”
The pipeline is expected to produce more novel new oral oncology drug approvals in 2017 than in 2016, with multiple approvals forecasted in breast cancer and blood cancers.
To view the report, visit http://bit.ly/2jnTRwl.
