 Although there are several different types of biological products, which the FDA notes includes proteins, monoclonal antibodies, and vaccines with little in common. For example, biologic drugs are large molecules typically manufactured from living cells and, therefore, are extremely difficult to produce or reproduce.
Although there are several different types of biological products, which the FDA notes includes proteins, monoclonal antibodies, and vaccines with little in common. For example, biologic drugs are large molecules typically manufactured from living cells and, therefore, are extremely difficult to produce or reproduce.
In fact, there can be variations of acceptable range within the same manufacturing process because biologics are processed using living cell-based technology and the make of these agents is not easily identifiable.
For this reason, they are also highly sensitive to storage and handling processes and typically have a shorter shelf life than your average oral medications. So, why research, develop, and ultimately manufacturer such complex, sometimes costly medications you might ask?
Tyrone’s Commentary:
We’ve seen this movie before, haven’t we? Much like small molecule generic equivalents, biosimilars are a big threat to biologic market share. It just takes time.
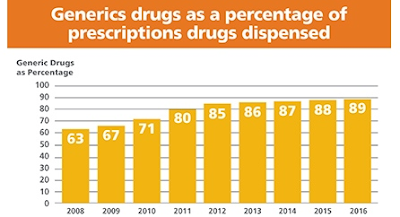 |
| Source: Generic Pharmaceutical Association annual savings and access report |
Because they are able to effectively treat a variety of medical conditions that previously had limited to no other treatments available, thereby changing the lives of patients with conditions such as rheumatoid arthritis, cancer, and anemia, just to name a few.
